MARLBOROUGH, Mass. and YOKNEAM ILIT, Israel, June 4, 2019 /PRNewswire/ -- ReWalk Robotics, Ltd. (Nasdaq: RWLK) ("ReWalk" or the "Company"), a leading manufacturer of robotic medical devices for individuals with lower limb disabilities, announced today that the U.S. Food and Drug Administration ("FDA") has cleared the Company's ReStore soft exo-suit system for sale to rehabilitation centers across the United States. ReStore is the only soft exo-suit with FDA clearance, and is intended for use in the treatment of stroke survivors with mobility challenges. Stroke is a leading cause of disability, which affects approximately 17 million people worldwide each year (1), and as many as 80% of people who have had a stroke will suffer from gait impairments. (2)
"The exo-suit achieves our commercial goal to offer a functional and affordable system that can be utilized in the 'Main Street' clinics in every community," said ReWalk CEO Larry Jasinski. "With a launch price of $28,900 as well as leasing options, ReStore offers cutting edge innovation with features that redefine therapy at a price that is accessable for a broader range of clinics than existing robotic technologies. The current gait training reimbursement codes enable immediate penetration and sales growth as part of our pathway to become a break even and profitable company."
In 2018, ReWalk launched a multi-center clinical study of the ReStore system across five of the country's leading rehabilitation centers. This data was submitted to the FDA in ReStore's 510(k) submission, and ReWalk expects to publish the results of the clinical study later this year.
"We are very encouraged by our initial experience and positive impact of ReStore in gait training for persons with stroke residual disabilities," said Moss Rehab's Chief Medical Officer Alberto Esquenazi, MD, who was one of the investigators in the multi-center trial. "By training the patient walking pattern in a more correct way, the expectation is that the brain will re-learn and better restore the walking function lost after a stroke."
The patented soft exo-suit technology was originally developed at Harvard University's Wyss Institute for Biologically Inspired Engineering where it also underwent initial clinical testing that demonstrated its potential to improve walking for stroke survivors. ReWalk and the Wyss Institute entered into a multi-year research collaboration agreement in 2016 which provides ReWalk access to future innovations that emerge from this collaboration and may be relevant to additional stroke products or other therapies.
"The idea of anchoring the body with textiles and flexible soft components is a fundamentally new way of applying assistance with a wearable robot," said Conor Walsh, a Professor of Engineering and Applied Sciences at the John A. Paulson Harvard School of Engineering and Applied Sciences and a Core Faculty member at the Wyss Institute for Biologically Inspired Engineering, who led a multi-disciplinary team to develop the soft exo-suit technology that has the ability to apply force to the body, but not restrict how a person moves. "This technology has broad potential, and we are currently testing additional concepts which can be applied to provide therapy and/ or mobility assistance for individuals with other diseases, such as multiple sclerosis and Parkinson's disease, and also potentially be used by a person at home and in their community," Walsh continued.
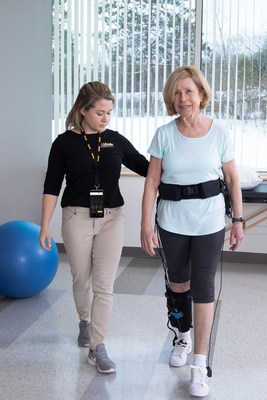
How it works: The ReStore system is comprised of a soft, garment-like design which connects to a lightweight waist pack and mechanical cables that help lift the patient's affected leg in synchronized timing with their natural walking pattern. The system provides targeted assistance to the patient during forward propulsion (plantarflexion) and ground clearance (dorsiflexion), two key phases of the gait cycle. The device also provides the physical therapists with extensive data during gait training with ReStore to inform strategies to optimize a patient's treatment and progress using real-time analytics.
ReStore is ReWalk's second major market segment, joining the ReWalk Personal 6.0—a robotic exoskeleton for home use by individuals with paralysis from a spinal cord injury—and the ReWalk Rehabilitation system, which allows for multiple patient uses through height and weight adjustments. "We believe the expansion to soft exo-suits gives ReWalk a diverse offering of innovative technologies and expands the Company's impact to millions of patients worldwide," added Jasinski. ReWalk intends to leverage its existing sales, training and service teams to commercialize the ReStore system. To date, ReWalk has placed nearly 550 exoskeletons in 26 countries and trained more than 270 rehabilitation clinics to conduct exoskeleton training worldwide.
(1) https://www.stroke.org.uk/sites/default/files/the_burden_of_stroke_in_europe_-_challenges_for_policy_makers.pdf
(2) https://synapse.koreamed.org/pdf/10.12786/bn.2017.10.e9
About ReWalk Robotics Ltd.
ReWalk Robotics Ltd. develops, manufactures and markets wearable robotic exoskeletons for individuals with lower limb disabilities as a result of spinal cord injury or stroke. The Company's mission is to fundamentally change the quality of life for individuals with lower limb disability through the creation and development of market leading robotic technologies. Founded in 2001, ReWalk has headquarters in the United States, Israel and Germany. For more information on the ReWalk systems, please visit www.rewalk.com.
ReWalk® is a registered trademark of ReWalk Robotics Ltd. in Israel and the United States.
Forward-Looking Statements
In addition to historical information, this press release contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, Section 27A of the U.S. Securities Act of 1933, and Section 21E of the U.S. Securities Exchange Act of 1934. Such forward-looking statements include those relating to projections regarding ReWalk's future performance and other statements that are not statements of historical fact and, in some cases, may be identified by words like "anticipate," "assume," "believe," "continue," "could," "estimate," "expect," "intend," "may," "plan," "potential," "predict," "project," "future," "will," "should," "would," "seek" and similar terms or phrases. The forward-looking statements contained in this press release are based on management's current expectations, which are subject to uncertainty, risks and changes in circumstances that are difficult to predict and many of which are outside of ReWalk's control. Important factors that could cause ReWalk's actual results to differ materially from those indicated in the forward-looking statements include, among others: ReWalk's ability to secure capital from its equity and debt financings in light of limitations under its Form S-3, the price range of its ordinary shares and conditions in the financial markets, and the risk that such financings may dilute ReWalk's shareholders or restrict its business; ReWalk's ability to maintain compliance with the continued listing requirements of the Nasdaq Capital Market and the risk that its ordinary shares will be delisted if it cannot do so; the risk of decreased liquidity in the market for ReWalk's ordinary shares and a reduced market capitalization of the Company following the recently-effected reverse share split, and the risk of dilution following the recently-effected increase in authorized share capital; ReWalk's expectations regarding future growth, including its ability to increase sales in its existing geographic markets, and to expand to new markets and achieve its planned expense reductions; the conclusion of ReWalk's management and the opinion of ReWalk's auditors in that there are substantial doubts as to ReWalk's ability to continue as a going concern; ReWalk's ability to maintain and grow its reputation and the market acceptance of its products; ReWalk's ability to achieve reimbursement from third-party payors for its products; ReWalk's limited operating history and its ability to leverage its sales, marketing and training infrastructure; ReWalk's expectations as to its clinical research program and clinical results; ReWalk's ability to improve its products and develop new products; ReWalk's ability to repay its secured indebtedness; the outcome of ongoing shareholder class action litigation relating to ReWalk's initial public offering; ReWalk's compliance with medical device reporting regulations to report adverse events involving its products and the potential impact of such adverse events on ReWalk's ability to market and sell its products; ReWalk's ability to gain and maintain regulatory approvals; ReWalk's expectations as to the results of, and the Food and Drug Administration's potential regulatory developments with respect to, ReWalk's mandatory post-market 522 surveillance study and ReWalk's 510k submission for the ReStore for stroke patients; ReWalk's ability to maintain adequate protection of its intellectual property and to avoid violation of the intellectual property rights of others; the risk of a cybersecurity attack or breach of our IT systems significantly disrupting our business operations; ReWalk's ability to use effectively the proceeds of offerings of securities; ReWalk's ability to establish a pathway to commercialize its products in China; the risk of substantial dilution resulting from the periodic issuances of ReWalk's ordinary shares; ReWalk's ability to maintain relationships with existing customers and develop relationships with new customers; the impact of the market price of ReWalk's ordinary shares on the determination of whether ReWalk is a passive foreign investment company; and other factors discussed under the heading "Risk Factors" in ReWalk's Annual Report on Form 10-K for the fiscal year ended December 31, 2018 filed with the U.S. Securities and Exchange Commission (the "SEC") and other documents subsequently filed with or furnished to the SEC. Any forward-looking statement made in this press release speaks only as of the date hereof. Factors or events that could cause ReWalk's actual results to differ from the statements contained herein may emerge from time to time, and it is not possible for ReWalk to predict all of them. Except as required by law, ReWalk undertakes no obligation to publicly update any forward-looking statements, whether as a result of new information, future developments or otherwise.

 View original content to download multimedia:http://www.prnewswire.com/news-releases/fda-issues-clearance-for-the-restore-exo-suit-the-first-soft-robotic-system-for-stroke-therapy-300861824.html
View original content to download multimedia:http://www.prnewswire.com/news-releases/fda-issues-clearance-for-the-restore-exo-suit-the-first-soft-robotic-system-for-stroke-therapy-300861824.html
SOURCE ReWalk Robotics Ltd.
Media Relations: Jennifer Wlach, E: media@rewalk.com, Investor Contact: Ori Gon, Chief Financial Officer, ReWalk Robotics Ltd., T: +972-4-9590123 , E: investorrelations@rewalk.com


![]() View original content to download multimedia:http://www.prnewswire.com/news-releases/fda-issues-clearance-for-the-restore-exo-suit-the-first-soft-robotic-system-for-stroke-therapy-300861824.html
View original content to download multimedia:http://www.prnewswire.com/news-releases/fda-issues-clearance-for-the-restore-exo-suit-the-first-soft-robotic-system-for-stroke-therapy-300861824.html